3D printing, biomaterials and enhanced regulatory pressures for Dental are coming
3D printing is now widely used across several industries, including healthcare and dental, where it can revolutionise many processes and services offered to patients, especially as new materials are being developed. In 2022, the European medical 3D printing market was estimated to be worth c. EUR 530m. In 2025 it is estimated to be around EUR 1bn with a fast growth rate. However, technical, business and safety challenges remain - hence the regulatory frameworks governing 3D technologies and resulting devices are crucial to ensuring their safety and efficacy. Yet it is increasingly hard for regulators to keep up with the pace of innovation, which results in unhelpful rigidities or grey zones, which then increasingly pressure dental manufacturers and clinicians.
DENTAL RESEARCHHEALTHCAREHEALTHCARE REGULATIONBUSINESS INNOVATION
Irene Petre
5/14/202612 min read
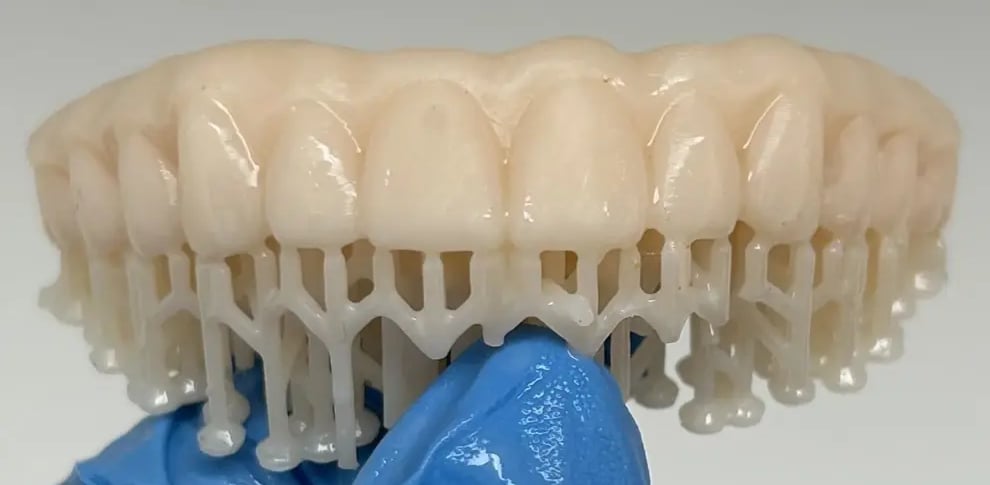
What is 3D printing?
Additive Manufacturing (AM) or 3D printing technologies have become pervasive across different sectors, including design, engineering, manufacturing - including biomedical – and they rely on digitally designed models to autonomously construct 3D objects. There are three elements of digital technologies that go hand in hand: (a) computer-aided design and manufacturing (CAD/CAM), increasingly AI driven, (b) digital data acquisition and (c) three-dimensional (3D) printing.
In the Dental field the adoption was first in implantology, in both surgical and prosthodontic stages and 3D printing has been used by some dental labs and dentists for the past 10-14 years, especially in countries like the US.
But is it really that 3D printing will replace subtractive manufacturing/computer numerical control (CNC) milling? Or are both going to co-exist for some time?
Despite the wide and successful adoption of CNC milling in the medical and dental fields for the past 30-40 years, including the dental implantology realm, it incurs high costs due to the wasted raw material in the process, besides the inevitable cutting tools wear, rendering them suitable only for a limited number of milling cycles, besides the inability to produce complex geometrical and hollow structures.
On the other hand 3D printing constructs objects via sequential material layering, following a digital 3D model, so it has the potential to create complex and hollow parts, besides being economical long term, efficient and more environmentally friendly.
One of the new strong materials used in 3D printing – but still debatable in dentistry – is titanium and titanium-allow objects. Some researchers claim that titanium and titanium alloy objects produced using 3D, are comparable to those created through traditional methods – but some people may be hypersensitive to titanium. But some scientists used 3D printing to fabricate porous Ti6Al4V dental implants, which were coated with a chitosan composite. This composite is non-toxic, promotes cell growth, and possesses strong mechanical properties, thereby facilitating the regeneration of bone.
Future developments in advanced biomaterials, artificial intelligence-assisted workflows, 4D bioprinting are expected to further expand the role of 3D printing in personalized and regenerative dentistry.
The Dental 3D Printing Market size is expected to grow from USD 3.7 billion in 2025 to USD 4.3 billion in 2026 and is forecast to reach USD 9 billion by 2031 at 15.9% CAGR over 2026-2031 according to Mordor Intelligence. By printing technology, stereolithography is the largest segment with 34.3% of the market in 2025 volume, but Digital Light Processing is forecast to grow the fastest at a 17.7% CAGR through 2031. For now dental laboratories are the largest 3D printing user segment globally with 59% of 2025 revenue, but clinics are expected to post a 20% CAGR through 2031.
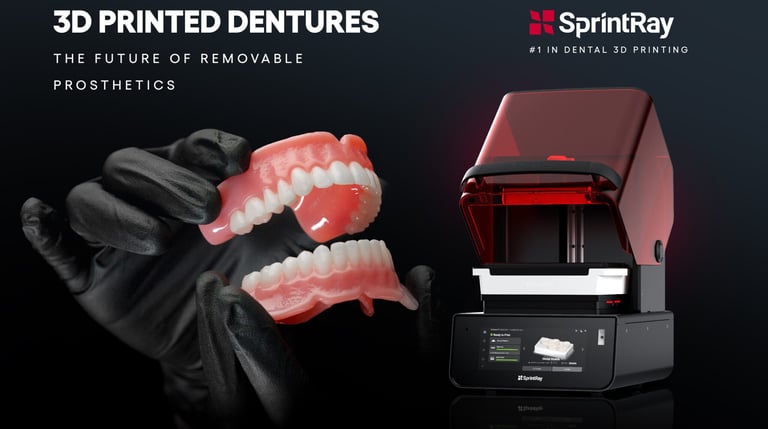
What are bioprinting and biomaterials
Biological 3D printing is an advanced technology that integrates cells, growth factors, and absorbable materials to fabricate intricate biological structures and there are three main techniques: inkjet bioprinting, extrusion bioprinting, and laser-assisted bioprinting.
Inkjet Bioprinting is lower cost and easily implementable, compatible with low-viscosity biomaterials and widely used in the field of cartilage tissue engineering.
Extrusion Bioprinting is high precision and compatible with a wide range of materials and because it does not require heating, it works well for printing structures that contain cells.
Laser-Assisted Bioprinting is high resolution and avoids possible problems like nozzle clogging and cell damage; it is also compatible with many biomaterials, but it is more expensive and less commonly used in cartilage engineering applications.
Biological 3D printing faces some challenges of course, such as the limited compatibility of bio-inks and the difficulty of replicating complex tissue structures with micron-level precision, but a newer method known as microsphere bioprinting is promising to eliminate some of these shortcomings.
Advancements in 3D-printable smart materials have brought about a new generation of "dimensional printing" such as 4D printing, which uses biomaterials to create pre-programmable products with the capacity for self-transformation in time - capable of changing shape over time in response to external stimuli in the environment such as temperature, pressure or moisture. For example, implants may become more flexible as surrounding tissues heal, therefore promissing to offer improved integration and comfort to the patient. Also 4D designs can improve load distribution and reduce stress concentrations, leading to longer-lasting implants that should (at least in theory) perform effectively under varying conditions.
Furthermore, materials used in 4D printing can be tailored to improve biocompatibility - for example creating bioactive coatings, porous structures for enhanced osseointegration and stimuli-responsive materials. It is worth mentioning that as research in 4D printing continues to advance, new materials, methods and devices are likely to be developed.
According to some sources such as Mordor Intelligence, resins represented in 2025 over 60% of the global 3D printing market by revenue. Although for many years they were deemed a bit too soft for permanent dental products, the recent development of durable, biocompatible, and highly specialised resins has expanded the possibilities in dental 3D printing — from same-day night guards to definitive crowns. Over the past five - six years, a lot was invested in resin innovation, at the beginning creating products which were used as provisional work but which are now used for definitive dental work.
According to producers like Sprint-Ray, new generations of 3D Printing materials now support higher-quality removable prosthetics (night guards, surgical guides, dentures, directly printed retainers and aligners) and, increasingly, fixed restorations such as crowns, bridges, inlays, onlays, veneers and full-arch hybrids. Additionally, selective laser melting and other metal 3D printing technologies allow for the production of titanium implants and cobalt-chrome partial denture frameworks.
According to the same firm - Sprint-Ray - and to other dental specialists, there is now a variety of biocompatible and flexible resins and high-strength ceramic and composite resins available; future innovation is looking at antibacterials and antifungal additives, eco-friendly resins (biodegradable and recyclable) and advanced light curable resins, with the aim of reducing production time and energy consumption.
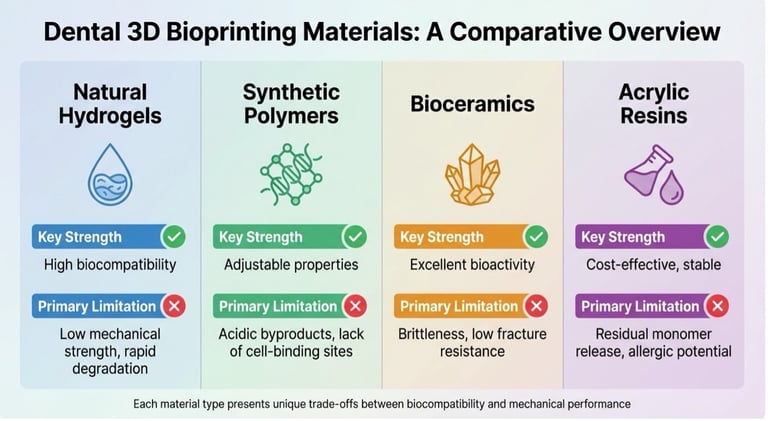
Source: Complete Smiles Bella Vista Dental Studio website (Australia), accessed 11th May 2026
Source: Sprint-Ray website, accessed 12th May 2026
Promises and challenges with 3D printing, bioprinting and biomaterials
3D printing is revolutionising the field of dentistry with three big promises for clinicians:
Increased speed of interventions by reducing processing waiting times for dental products and getting to same-day restorations (doing everything chair-side)
More autonomy and (quality) process control
Long term cost reduction.
As the technology evolved significantly over the past 10 -15 years, printing itself (which is only part of the process with pre-processing and post-processing and finishing taking a lot more time) has indeed reduced from circa 8-9 hours c.12 years ago to circa 8-9 minutes today, depending on the printer, skill of the operator and what is being printed. However a few hurdles remain which are technical on one hand, related to material or process shortcomings and on the other hand are business related given increased specialist training required, high investment costs and increasing regulatory burdens. The regulatory are is becoming a new type of challenge altogether, especially with increasing complexities of ISO standards and of regulations such as the MDR/IVDR, GDPR and AI Act in the EU.
Technical challenges
Although 3D printing materials have advanced significantly, not all dental materials are available in printable form. Traditional restorations, such as zirconia and lithium disilicate crowns are still primarily milled because ceramic 3D printing technology is still developing. Some resins used for 3D-printed restorations lack the durability and wear resistance of conventional materials used for long-term restorations and although producers claim improved performance in new generation materials, the truth is only time can tell.
Milled restoration often require minimal post-processing but 3D printed dental products need additional finishing before they can be used, which can include curing, sintering, polishing, surface treatments etc. depending on the material and printing method used. Continuous Liquid Interface Production (CLIP) now alows for faster printing, without visible layer separations and is used for crowns, aligners or dentures. Litography-Based Ceramic Manufacturing (LCM) is now used for Zirconia printing for crowns and bridges – these products are meant to be more durable and esthetic but their endurance will only be tested in time.
Business challenges
Good quality dentistry and dental products pay particualr attention to detail and sit between art and science. Training was always involving in terms of time and financial investment required. But with the adoption of fast evolving digital and AI workflows and 3D physical technologies, those efforts are set to double. Not only need newly trained clinicians learn all the medical science behind but they also need to become tech experts, especially if they aspire to also adopt 3D printing in their own studios. They need to have improved knowledge of materials, including new materials, chemical reactions etc. but also need to get versed in using new fast evolving digital technology. Paying for training it is not enough – they need many hours of practice in order to develop the right level of skill for pre-processing, post-processing and finishing requirements. Even if expert 3D printing users, these activities take a long time. There is also the issue of lack of availability of training in these new technologies and materials in many parts of the the world – including in Europe, particularly in smaller towns and rural areas.
The high initial investment in technology and software packages is another problem, especially for smaller or younger dental clinics – investing in an industrial grade printer, CAD/CAM software and associated training can cost many tens of thousand EUR, if not hundreds, depending on what clinicians aspire to be able to do. A cheap 3D printer can now be acquired on Amazon for prices as low as EUR 500-1000 but there are two main issues with low cost products: one one hand their abilities are not going to be comparable to more high-end printers but it is a problem of skill as well: more skill and more work is required in post-production and polishing of products obtained with a less expensive but less performant 3D printer.
High-end SLM, SLA, and ceramic 3D printers cost tens of thousands of dollars. In term of bioprinting - entry-level bioprinters, such as the Tissue Scribe, start at around EUR 1,500 according to some sources, while mid-range models like the Lulzbot Bio are priced at roughly EUR10,000. However, professional-grade automated systems can climb in the hundred of thousands of EUR or USD.
Of course material costs add another layer of expense - especially for bioprinting. Unlike standard 3D printing filaments, bioinks are far more complex, as they must include living cells and bioactive agents. Knowledge about the properties of these materials, bio-security, knowledge of providers etc. are therefore mandatory.
So there comes another operational problem in dental studios: who in the team is going to learn to do what, who is going to take care of which steps of the process and get really skilled at it? Who is going to be responsible for sourcing the materials, software, 3D printers, scanners etc? Sourcing from one provider or different ones? Who is going to be responsible for post-production? Etc.These processes are complex and there is no margin for error, so it becomes harder for one clinician to have the time and ability to master all of them. Team cohesion and granular specialisation then become very important.
Potential safety issues and regulatory responses
3D printed dental products are placed inside the human body, so they need to meet stringent FDA, CE and ISO certifications, to make sure they are safe for long term use and do not cause adverse reactions and complications for human health. Cross-contamination is one of the key risks that needs to be minimised through strict sterilisation.
Additionally, there can be cytotoxicity and biocompatiblity concerns regarding new materials such as antibacterial and antifungal addititives used in 3D bioprinting, as well potential issues around mechanical stability, and material integrity as Incorporating additives often leads to reduced mechanical strength, lower flexural strength, and reduced wear resistance. The addition of nanoparticles or other agents can lead to higher porosity, making the printed material too brittle for load-bearing applications like crowns or implants.
Overall there are several biocompatibility and durability problems with many materials – but a key issue with dental 3D printing materials is residual monomer toxicity. During the printing process, incomplete polymerisation of substances like resins can leave behind harmful substances like methyl methacrylate, which has been linked to mucosal irritation, allergic reactions, and pulp damage. Also to avoid safety problems, bioinks are made with biocompatible and non-toxic cells and biomaterials and undergo thorough biological testing, including both in vitro (lab-based) and in vivo (animal or human-based) assessments.
But these potentially toxic substances in post-printing processes cannot be completely eliminated and they can affect the health of technicians and clinicians but also trigger reactions within patients if not handled with care or if the quality of materials is low.
These are just some of the potential safety complications - overall there is still a lack of standardisation in the regulation and testing of many products and materials or at least many grey zones, in both existing products and technologies and especially surrounding ong-term clinical use of new ones such as bioinks or the antimicrobial-loaded 3D printed materials.
Also in terms of material certification there is significant regulatory variability across geographies. Clearance takes 6-9 months under the FDA’s pathway in theory but 18-24 months in the EU, where MDR requires full clinical-evaluation reports. China’s NMPA upgraded long-term dental resins to Class III in 2024, aligning them with implant devices and delaying market entry by a year. Japan’s PMDA requires cytotoxicity assays on five-year-old specimens, whereas Brazil’s ANVISA requests real-time stability data, forcing suppliers into redundant test cycles. So smaller formulators lacking regulatory expertise and staff can lose market share to larger firms that amortise compliance costs across larger portfolios.
But as innovation grows and the variety of new materials, production technologies and end products is increasing in dentistry, regulations in many regions are trying to catch-up.
The EU for example has a rather strict approach regarding "patient-matched" or "personalised" dental prints under the EU Medical Device Regulation (EU MDR 2017/745), which became applicable in May 2021. Under the regulation many devices previously considered "custom-made" are now classified as "patient-matched" (or "adapted") devices, meaning they must comply with more stringent regulatory requirements, including formal conformity assessments.
One part of the MDR’s pre-market regulation, called "Custom Device Exemption", allows certain devices made per a written description by an authorised person to bypass some requirements, like CE marking, but must still comply with quality management and other regulatory obligations. However the MDR states that ‘patient-matched’ devices (for a specific patient) produced through a reproducible process do not qualify for Custom Device Exemption and must follow a stricter regulatory pathway.
In the MDR, drawing the distinction between ‘patient matched’ and ‘mass-customised’ devices (which are prescribed generically, and then adapted to individuals) is still a grey area for 3D printed medical devices.
The MDR also provides a regulatory exemption for in-house, non-industrial manufacturing of 3D printed devices within healthcare institutions (such as hospitals), known as the Health Institution Exemption. But it is unclear how how might it cover an industrial partner based at a hospital or large clinic.
Post-market surveillance is more rigorous in the EU compared to other regions like the US, where, for example, compliance with pre-market regulations might provide a full defence against liability claims, but this is only partial in Europe. under the MDR, product liability for 3D printed devices is currently unclear due to the boundary between medical negligence and product liability being ‘blurred’ and complicated by a potentially ‘decentralised production model’ (where for example a healthcare specialist could have designed and use a certain device but also use 3rd party manufacturers and inhouse hospital manufacturers) and the many different parties that could be involved in delivering these devices to patients.
The new EU Product Liability Directive (PLD, 2024) proposed changes expand the definition of 'product' to include software, digital files and services, broadening the scope of liable parties.
And another grey area is around the lack of current intellectual property (IP) legislation surrounding the computer aided design (CAD) files that are the blueprints used to manufacture 3D printed medical devices.
The implication is that small companies or institutions might find it challenging to navigate the complex regulatory scene, especially when producing tailored 3D printed devices. Small dental labs, dental studios and especially material manufacturers must at least have a Quality Management System (QMS) in place and potentially involve a Notified Body (third-party auditor) to obtain a CE mark for their devices.
The need for extensive documentation, validation and quality management systems could slow down the time-to-market for innovative solutions.
The EU uses a staggered transition for "legacy devices" (those certified under the old MDD) that extends until 31 December 2027 for high-risk (Class III/IIb) devices and 31 December 2028 for lower-risk devices (Class IIa/I) and starting May 2026, manufacturers must register devices in EUDAMED (the EU database) and comply with Unique Device Identification (UDI) requirements, bringing them into more scrutiny.
The lack of regulatory clarity is also coupled with a lack of availability of training in these new technologies and materials in many parts of the the world – including in large cities in Europe, not just in rural areas.
Regulatory pressures are not unique to Europe though. In Australia for example any personalised 3D prints, once considered "custom-made", are now classified as "patient-matched medical devices" (PMMDs). These devices must be listed in the Australian Register of Therapeutic Goods (ARTG) by 1 July 2029 - this brings new legal responsibilities for clinicians, including when importing bioprinting materials like resins or bioinks from overseas.
Regulatory pressures are unlikely to deter technicians or dental studios from adopting 3D printing and bioprinting. It is most likely they will change their buying and partnerships behaviour in order to share risks or eliminate certain risks altogether. New business models and partnerships models are and will continue to emerge in the Dental ecosystem in the coming years.
Some sources:
https://www.sciencedirect.com/science/article/pii/S2405886624000289
https://www.sciencedirect.com/science/article/abs/pii/S2405886625000223
https://sprintray.com/innovations-in-3d-printing-materials-for-dental-applications/
https://completesmilesbv.com.au/challenges-dental-3d-bioprinting/
https://www.mdpi.com/2073-4360/16/24/3550
https://www.sciencedirect.com/science/article/pii/S2472630322011207
https://health.ec.europa.eu/medical-devices-topics-interest/custom-made-devices_en
https://www.mordorintelligence.com/industry-reports/dental-3d-printing-market
IGEA Healthcare
Strategic Advisory for Life Sciences
Switzerland, UK, Italy
contact@igeahealthcare.com
© 2025 - 2026. All rights reserved.
